 Atomic Radius Trend in Periodic Table (Simple Explanation). These factors are interdependent and determine the overall size of an atom. 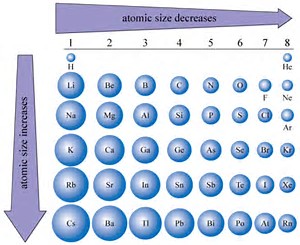
The factors affecting atomic radius are nuclear charge, electron configuration, and distance of the valence electrons from the nucleus. Experimental methods, such as X-ray crystallography, electron diffraction, and spectroscopy, are used to measure atomic radius. 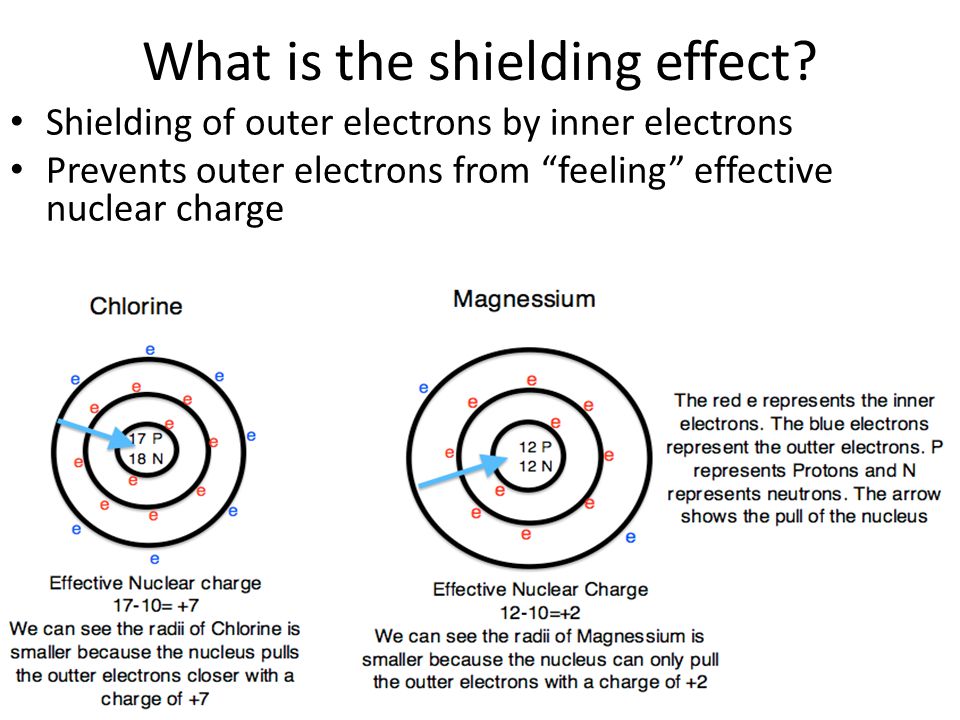
It helps us understand how atoms interact and affect their chemical properties. o Lanthanides and actinides (the two rows of elements below the main table) have. By analyzing the wavelengths of light that are absorbed or emitted by an atom, scientists can determine its atomic radius.Ītomic radius is the size of an atom, measured in picometers, from the center of the nucleus to the outermost electron in the electron cloud. electronegativity, ionization energy, electron affinity, atomic radius. Spectroscopy involves studying the interactions between light and matter.As the electrons pass through the film, they scatter off the atoms, creating a diffraction pattern that can be analyzed to determine atomic radius. In electron diffraction, a beam of electrons is directed at a thin film of the element being studied.The X-rays diffract off the atoms in the crystal, and the resulting diffraction pattern can be used to determine the distance between the atoms and thus their radii. In X-ray crystallography, a beam of X-rays is directed at a crystal of the element being studied.These methods involve studying the behavior of atoms when they interact with certain forms of energy, such as X-rays or electrons. Atomic radius is typically measured using various experimental methods such as X-ray crystallography, electron diffraction, and spectroscopy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
